About US

Leading the Future of Medical Aesthetics through Innovative Silk Technology,
Technology Dersmithson.
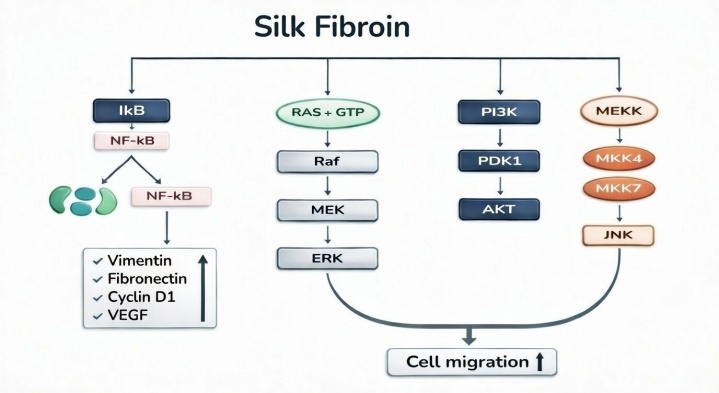
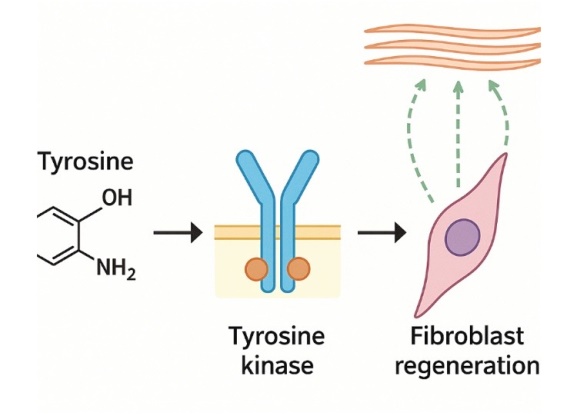
Dersmithson is an R&D specialized enterprise dedicated to developing high-end composite fillers. We combine "Silk Fibroin"—the most noble natural protein discovered by humanity—with next-generation polymer materials, utilizing our proprietary extraction technology. Our journey began on the global stage, connecting Korea, China, and the United States under the name Maxgen. Over the years, our original technologies have been recognized by numerous global partners, leading to successful patent registrations and technology transfers. We have consistently spearheaded innovation in the field of biomaterials.
Today, we are embarking on a second leap forward as Dersmithson, headquartered in Poland, the heart of Europe. Dersmithson's composite fillers focus on more than just enhancing aesthetic appearance; we are committed to achieving the safest and most natural biocompatibility for the human body. By integrating the technological assets accumulated through Maxgen with Dersmithson's sophisticated research capabilities, we provide premium solutions that medical professionals and clients worldwide can trust.
We believe that the value of technology truly shines when it enriches human life. Through continuous R&D investment and rigorous quality control, we promise to set a new global standard in the medical aesthetic market.
Thank you.
CEO, Dersmithson